Peripheral neuroma remains a worldwide medical challenge, often leading to persistent mechanical allodynia. Currently, there is a lack of ideal clinical treatments. Although nerve conduit technology has shown potential in treating residual neuromas, the recurrence of neuromas remains prominent due to the inability to continuously and effectively inhibit pathological vascular regeneration in the nerve stump. Therefore, there is an urgent need to develop more precise and efficient intervention strategies.

Recently, the team led by Yongwei Pan, Deputy Director of the Department of Orthopedics and the Sports Medicine Center at Beijing Tsinghua Changgung Hospital, in collaboration with Xiumei Wang's team from the School of Materials Science and Engineering at Tsinghua University, proposed a new strategy to prevent neuroma formation by continuously and locally antagonizing vascular endothelial growth factor receptor 2 (VEGFR2) within a spatially confined nerve end-cap, thereby blocking pathological vascular regeneration (Figure 1). The research team employed 3D printing technology to arrange biofunctional GelMAMAVP microspheres in an ordered array within the spatially confined nerve conduit, coordinating structural mechanics with the dual signals of targeted vascular inhibition. This provides a new approach for the treatment of traumatic painful neuromas.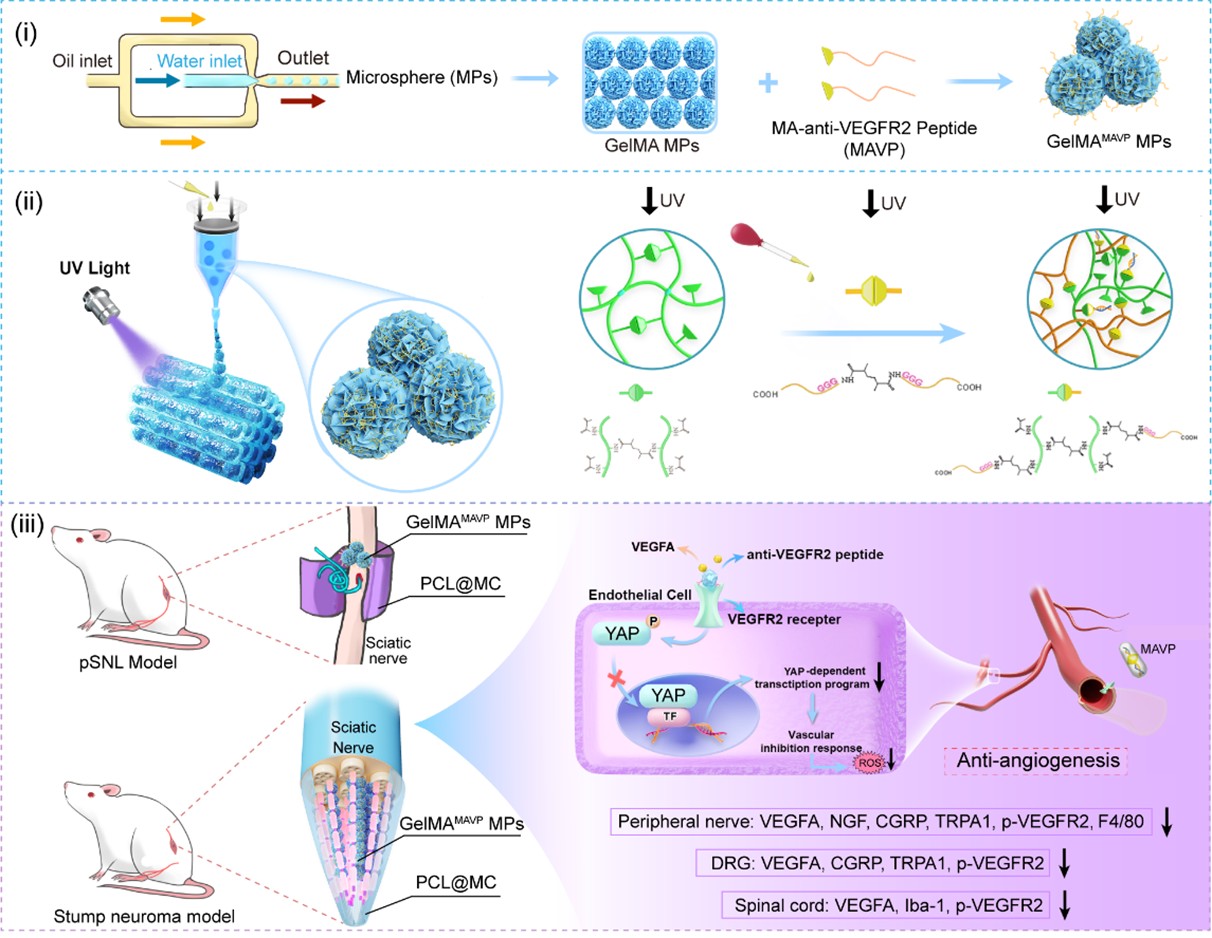
Figure 1. Fabrication of GelMAMAVP MPs using microfluidics and bioprinting technology to continuously regulate the residual nerve microenvironment and alleviate pain by targeting VEGFR2 antagonism.
This study builds upon the team's previous research. The two teams had earlier developed a spatially confined nerve conduit and revealed its mechanism of regulating nerve growth and inflammatory microenvironment through the YAP mechanotransduction pathway (Nature Communications, 2025, 16(1):66). Based on these findings, the research team further introduced localized vascular inhibition signals, significantly improving allodynic behaviors—including mechanical, cold, and thermal pain, as well as gait abnormalities—in a peripheral nerve pain hypersensitivity model (Figure 2).
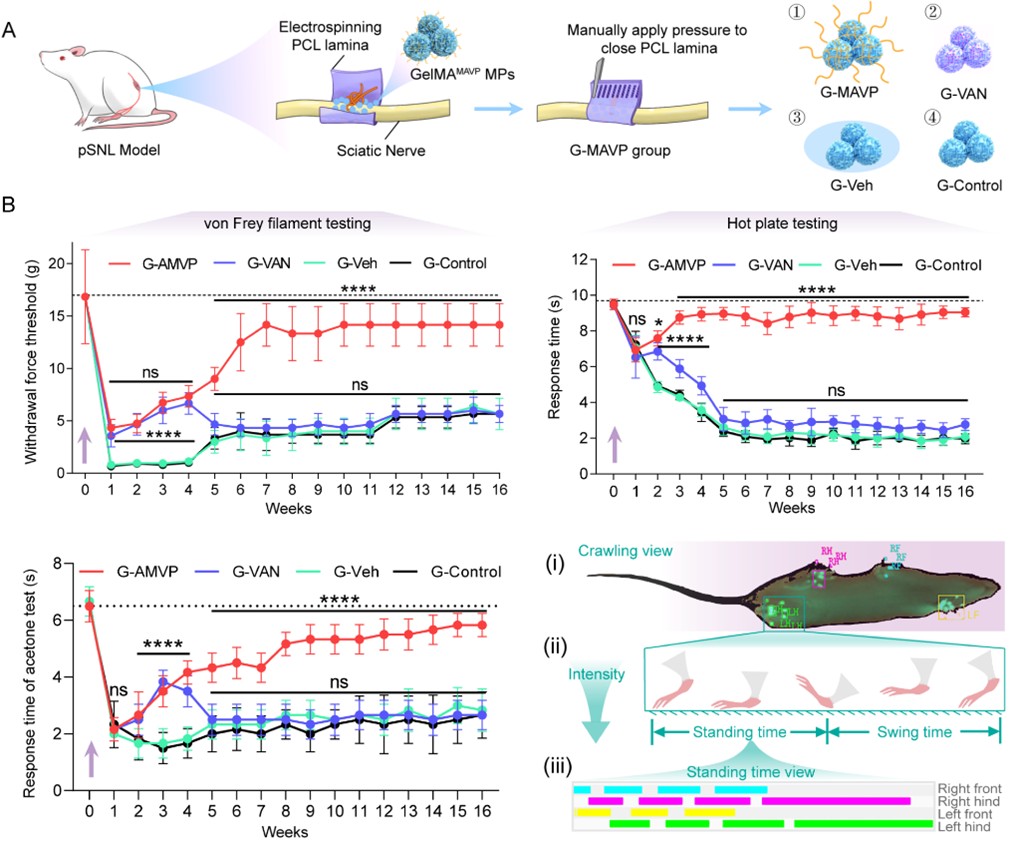
Figure 2. GelMAMAVP MPs significantly alleviate pain behaviors in a mechanical allodynia model.
Mechanistic studies revealed that in a rat model of painful neuroma, targeted VEGFR2 antagonism further slowed axonal growth, inhibited reactive oxygen species release in vascular endothelial cells, reduced scar tissue hyperplasia, and indirectly decreased the expression of pain-related proteins (CGRP and TRPA1) in the dorsal root ganglia (DRG). It also suppressed microglial activation in the spinal dorsal horn, thereby significantly attenuating pain signal transmission in the central nervous system (Figure 3). By precisely modulating the residual nerve microenvironment, this strategy not only opens a novel targeted pathway for treating traumatic painful neuromas but also lays an important foundation for understanding the mechanisms underlying chronic pain after peripheral nerve injury, demonstrating significant potential for clinical translation.
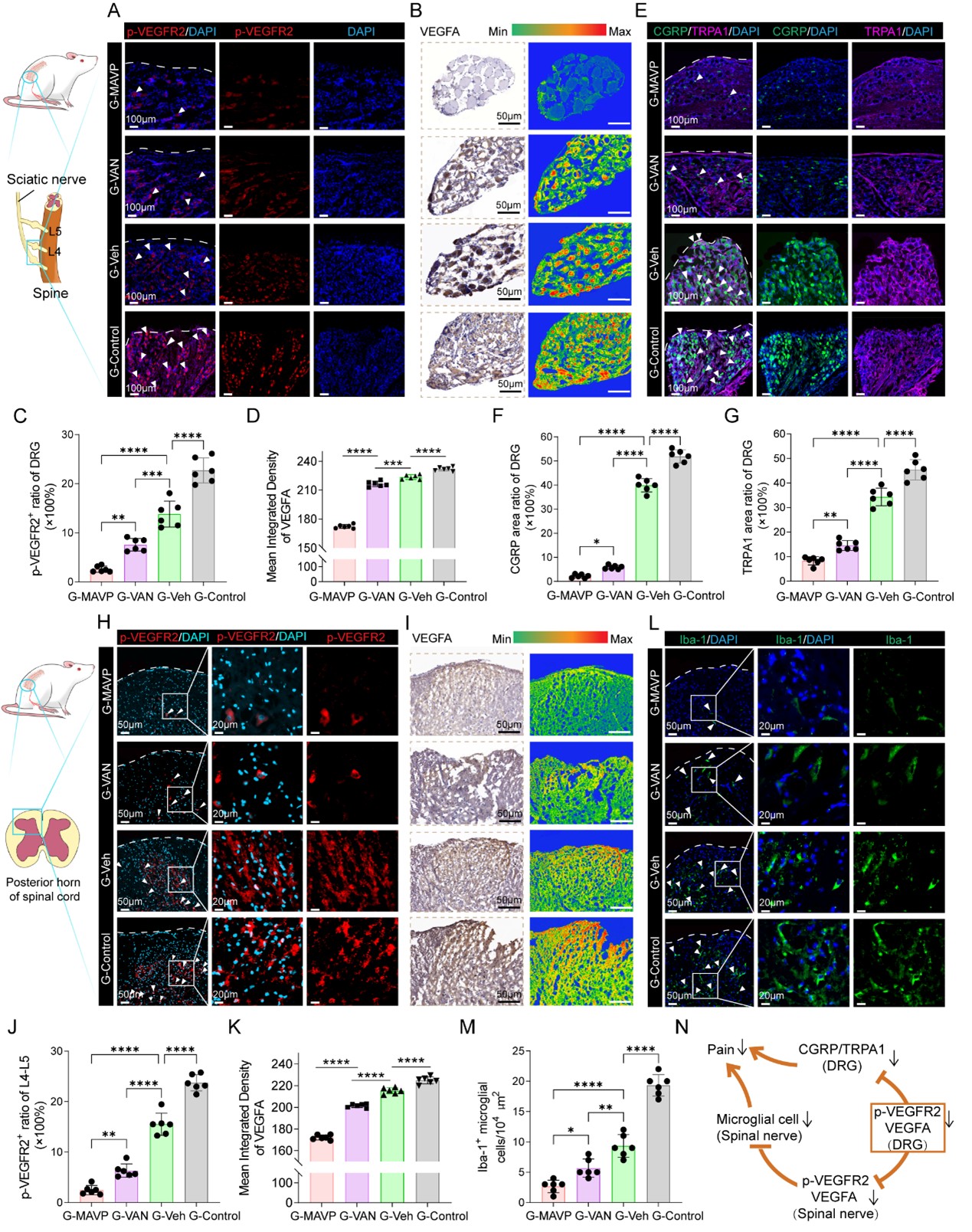
Figure 3. Spatially confined nerve conduits loaded with GelMAMAVP MPs effectively inhibit central pain signaling in a neuroma model.
The findings have been published in the journal Bioactive Materials under the title "Targeting VEGFR2 inhibition within a spatially-confined conduit promotes nerve self-resolution and alleviates mechanical allodynia." Yongwei Pan and Xiumei Wang are the corresponding authors. Co-first authors include Xiaobin Luo (Tsinghua Shuimu Scholar Postdoctoral Fellow), Toshitatsu Nagayasu (master's student at the School of Materials Science and Engineering, Tsinghua University), and Yuhao Zhang (Ph.D. student at the School of Clinical Medicine, Tsinghua University/Beijing Tsinghua Changgung Hospital). This work was supported by the Beijing Science and Technology Project, the National Key Research and Development Program of China, the National Natural Science Foundation of China, and the Beijing Natural Science Foundation.
More: https://www.sciencedirect.com/science/article/pii/S2452199X26001398
Author: Xiaobin Luo
Reviewed by: Yongwei Pan and Xiumei Wang